Mechanistic study of titania-based CO2 photocatalysis
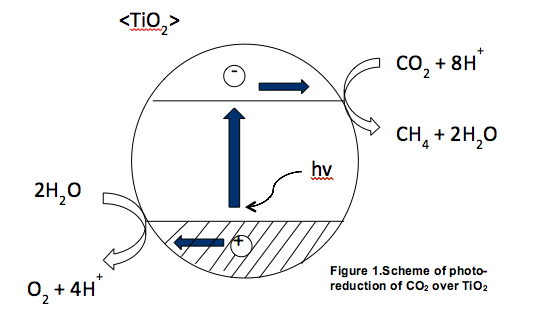
The reduction of CO2 emissions to the atmosphere is one of the major environmental issues we have to solve. One attractive scenario is to convert unwanted CO2 into fuels. However, the low efficiency of photocatalysts prevents it from being utilized at large scale. The major bottleneck of designing novel catalyst is lack of knowledge in mechanism of photocatalytic CO2 reduction. By combining theory and experiments we try to understand the mechanism of CO2 adsorption and activation on photocatalysts.
Our current focus is on the adsorption of CO2 on titania based catalyst. By modifying the titania surface with different basic oxides, we try to demonstrate how the adsorption of CO2 affects the overall reactivity. Atomic layer deposition (ALD) is used to synthesize thin oxide layer on titania. We use in situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) to characterize the synthesized catalysts and to investigate the CO2 adsorption on the surface. Periodic density functional theory (DFT) calculations are performed to model the surface structure of the catalyst and to study CO2 adsorption. For future work, the photocatalytic activity will be measured and will be correlated with the CO2 surface coverage.
References:
(1) Fujishima, A.; Honda, K. Nature, 1972, 238, 37.
(2) Danon, A; Stiar, P.; and Weitz, E. Catal. Lett. 2011, 141, 2057
(3) Henderson, M. Surface Science, 1998, 400, 203.
(4) He, H.; Zapol P.; Curtiss, L. J. Phys. Chem. C 2010, 114, 21474
Grant: ICEP

